In recent years, the rising levels of nitrate contamination in water sources have become a pressing environmental and public health issue. As industrial activities and agricultural runoff increase, the risk of nitrate entering drinking water supplies poses significant dangers to human health, especially for vulnerable populations like infants, leading to conditions such as methemoglobinemia, or “blue baby syndrome.” Tackling this issue requires innovative solutions that prioritize not only effectiveness but also sustainability. Recent breakthroughs from Yale University, particularly through the work of Lea Winter and her team, promise a transformative approach to nitrate removal that could significantly shift current paradigms.
The Traditional Approach: Limitations and Challenges
Historically, the removal of nitrates from water has relied on two primary methods: separation and destruction. While separating nitrates might seem like a straightforward solution, it often results in a concentrated waste stream that poses a risk of re-entry into the ecosystem. Conversely, destruction methodologies, such as biological denitrification, are frequently employed in wastewater treatment but come with their own set of challenges. These methods are sensitive to environmental conditions, and even minor fluctuations in pH, temperature, or microbial health can jeopardize the entire process. Moreover, biological systems are inherently slow, defeating the urgency required to address contamination effectively.
The scientific community has turned to electrocatalytic processes as a faster alternative. These techniques enable more controlled environments for nitrate destruction. However, conventional electrochemical methods are fundamentally limited by their reliance on flat plate electrodes that inhibit the efficient transport of nitrate—creating a bottleneck in processing speed. The complex interplay between reaction rate and nutrient transport results in inefficiencies that have long hampered these systems.
Innovative Solutions: Electrified Membranes
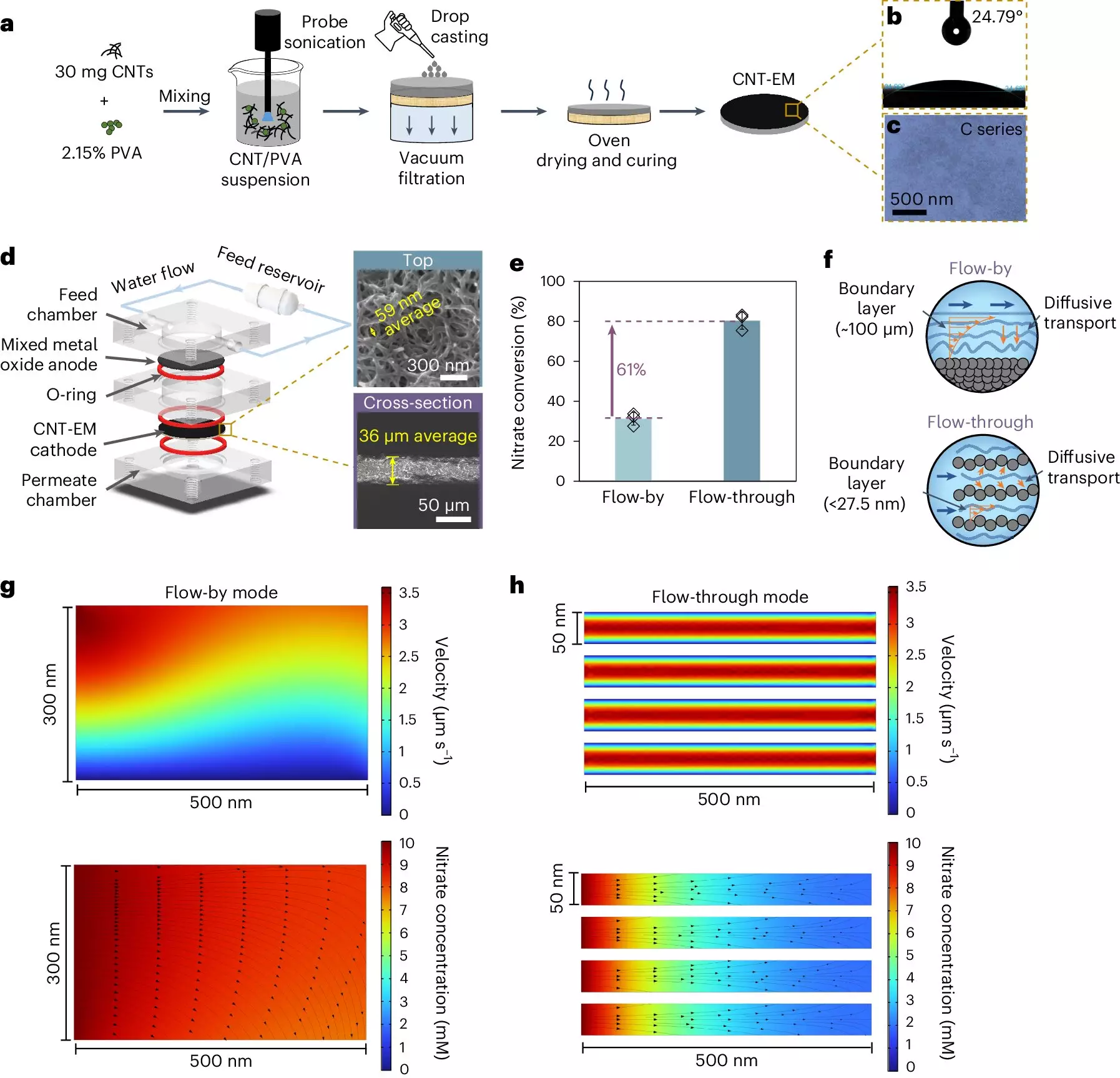
Enter Lea Winter’s groundbreaking research. Instead of relying purely on traditional systems, her laboratory has developed electrified membranes composed of carbon nanotubes, which radically transform the nitrate removal process. This innovative design takes advantage of nanoscale pores within the membrane that are a mere 50 nanometers wide—significantly smaller than the 100 micrometers found in conventional systems. The reduced boundary layer drastically accelerates the movement of nitrate, facilitating quicker reactions at the electrode surface.
This shift in the physical structure of membranes not only addresses the limitations of traditional electrochemical setups but also promotes the efficient conversion of nitrate without the use of metal catalysts. As a result, Winter’s membranes can achieve nitrate destruction at rates comparable to or exceeding those of metal-based systems. This advancement optimizes performance in a way that has the potential to reshape the future of water treatment practices.
Speeding Up Nitrate Removal: A Game Changer
One of the most striking features of this new technology is its incredible efficiency. Conventional electrochemical systems can take hours to achieve satisfactory nitrate removal—often requiring extensive time to contact all nitrate present within the water. However, with Winter’s innovative system, the notable reduction in nitrate concentration can occur in a matter of seconds. Early tests have shown that nitrate removal can reach levels of 80% within merely 15 seconds. Such remarkable speed not only enhances the safety of our water supply but also paves the way for real-time remediation solutions that could be deployed in urgent situations.
This leap in efficiency is particularly valuable in areas dealing with lower concentrations of nitrates that are typical of much of the contaminated water found in the environment today. By demonstrating versatility across varying nitrate levels, Winter’s membranes could serve as a definitive solution for municipalities facing mounting water safety challenges.
A Real-World Application: Testing in Natural Waters
To assess the practicality of her innovations, Winter and her team conducted real-world tests using water samples from Lake Wintergreen—a prominent location near the Yale campus. The impetus behind this experimentation was to not only validate the technology in controlled conditions but to explore its feasibility in treating typical contamination scenarios. These initial trials represent a crucial step in translating laboratory advancements into real-world applications that could ultimately protect and improve the quality of our vital water sources.
As these innovative technologies continue to evolve, they could lead to a new era of water treatment practices, prioritizing not just removal, but the complete destruction of nitrate contaminants. The implications of such research extend far beyond academia, promising a healthier future for communities impacted by nitrate pollution. This revolutionary approach heralds hope in our fight against pervasive environmental threats, showcasing the power of interdisciplinary collaboration to address some of the most pressing challenges of our time.

Leave a Reply