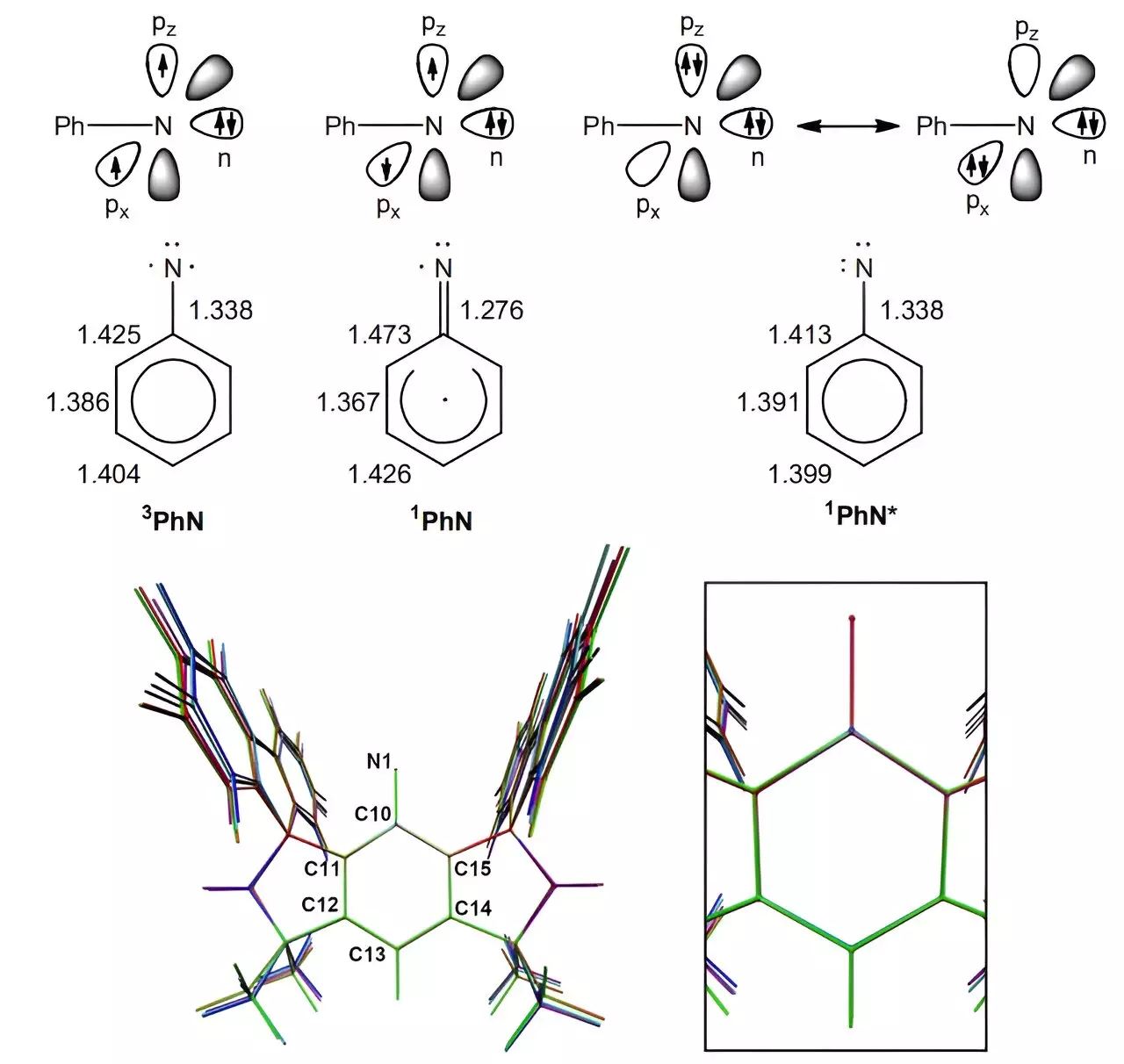
In a groundbreaking study published in the journal Science, a team of chemists at the University of Bremen in Germany has made a significant advancement in nitrene chemistry. Nitrenes, which are reactive intermediates and analogs of carbenes, have traditionally been known for their fleeting nature due to their short reaction times, typically in the nanoseconds. However, this team of researchers has successfully developed a new type of nitrene that exhibits slow reactions lasting for up to three days, opening up a realm of possibilities for their application in various fields.
To achieve this groundbreaking feat, the researchers utilized a chemical scaffolding known as MSFluind, which was first introduced in 2011 by another research team. This innovative scaffolding has shown promise in stabilizing various reactive molecules, including nitrenes. By employing MSFluind, the researchers were able to impede access to the nitrogen atom in a reaction, leading to a significantly decelerated process. Through exposure to ultraviolet light, the team was able to reduce an azide precursor to form the slow-reacting nitrene, MSFluindN. Subsequent analysis using X-ray crystallography, electron paramagnetic resonance spectroscopy, and superconducting quantum interference device magnetometry confirmed the persistence of the nitrene in its spin-triplet ground state for up to three days.
This groundbreaking discovery paves the way for a new class of nitrenes that could potentially revolutionize the field of chemistry and catalysis. The ability to manipulate and stabilize nitrene reactions opens up exciting opportunities for the synthesis of new transition metal complexes, which were previously challenging to achieve due to the transient nature of nitrenes. The researchers’ innovative approach not only provides a deeper understanding of nitrene characteristics but also enables the exploration of novel applications in various industrial processes.
Future Directions in Nitrene Chemistry
Moving forward, the research team plans to further investigate the potential uses of slow-reacting nitrenes in catalytic reactions and organic synthesis. By harnessing the unique properties of these stabilized nitrenes, chemists can unlock new pathways for the development of advanced materials and pharmaceutical compounds. The unprecedented longevity of nitrene reactions achieved in this study marks a significant milestone in nitrene chemistry and sets the stage for continued innovation in the field.
The development of slow-reacting nitrenes represents a paradigm shift in the field of chemistry, offering new possibilities for molecular design and catalysis. The research conducted by the team at the University of Bremen not only expands our understanding of nitrene reactivity but also opens doors to novel applications with far-reaching implications. The future of nitrene chemistry is brighter than ever, thanks to this groundbreaking discovery.

Leave a Reply